You’ve probably heard that generic drugs are cheaper versions of brand-name medications. But have you ever wondered why some patients stick with the brand even when a generic is available, or why they might choose a specific type of generic over another? The answer often lies in a lesser-known category called authorized generics, which are identical copies of brand-name drugs produced by the same manufacturer but sold under a different label. These drugs sit in a unique middle ground between expensive brand names and standard generics, influencing patient choices in surprising ways.
What Exactly Are Authorized Generics?
To understand why patients choose them, we first need to define what they are. An authorized generic is not your typical off-brand medication. It is chemically identical to the brand-name drug, including both active and inactive ingredients. The key difference? It lacks the brand name on the label.
Unlike traditional generics, which must go through a rigorous process called the Abbreviated New Drug Application (ANDA) to prove they are bioequivalent to the brand, authorized generics bypass this step. They are manufactured by the brand company itself using the existing New Drug Application (NDA). This means they come from the same factory, use the same raw materials, and follow the same production protocols as the original brand-name product. The Federal Trade Commission (FTC) has tracked these products since at least 2001, noting they serve as a strategic tool for brand manufacturers to maintain market share after patent expiration.
| Feature | Brand-Name Drug | Authorized Generic | Traditional Generic |
|---|---|---|---|
| Manufacturer | Original Brand Company | Original Brand Company | Third-Party Competitor |
| Regulatory Pathway | New Drug Application (NDA) | Existing NDA | Abbreviated New Drug Application (ANDA) |
| Ingredients | Proprietary Formula | Identical to Brand | Bioequivalent Active Ingredients; Different Inactives |
| Price Reduction vs. Brand | N/A | 4-18% | 30-50%+ |
| Market Entry Timing | Patent Protected | During 180-Day Exclusivity Window | After Patent Expiration/Challenge |
The Patient Experience: Why Formulation Matters
When it comes to patient preferences, trust and consistency play huge roles. Many patients worry that switching to a generic might change how they feel because of differences in inactive ingredients-things like fillers, binders, or dyes. While traditional generics must demonstrate bioequivalence, meaning they deliver the same amount of active ingredient into the bloodstream, they can still contain different inactive components.
A landmark 2018 FDA-sponsored study published in the BMJ analyzed 210,000 patient records and found something significant. Patients who switched from a brand-name drug to an authorized generic had a "switchback" rate of only 22.3%. In contrast, those who switched to traditional generics had a switchback rate of 28.7%. A switchback happens when a patient returns to the brand-name drug, often due to perceived side effects or dissatisfaction. This 6.4% difference suggests that patients perceive authorized generics as more similar to their trusted brand product, likely because the formulation is truly identical.
In real-world scenarios, this matters. If you take a medication for a sensitive condition like heart disease or mental health, even minor changes in how a pill dissolves or interacts with your stomach lining can cause discomfort. Authorized generics eliminate this variable entirely. You get the exact same physical product, just without the premium price tag associated with the brand logo.
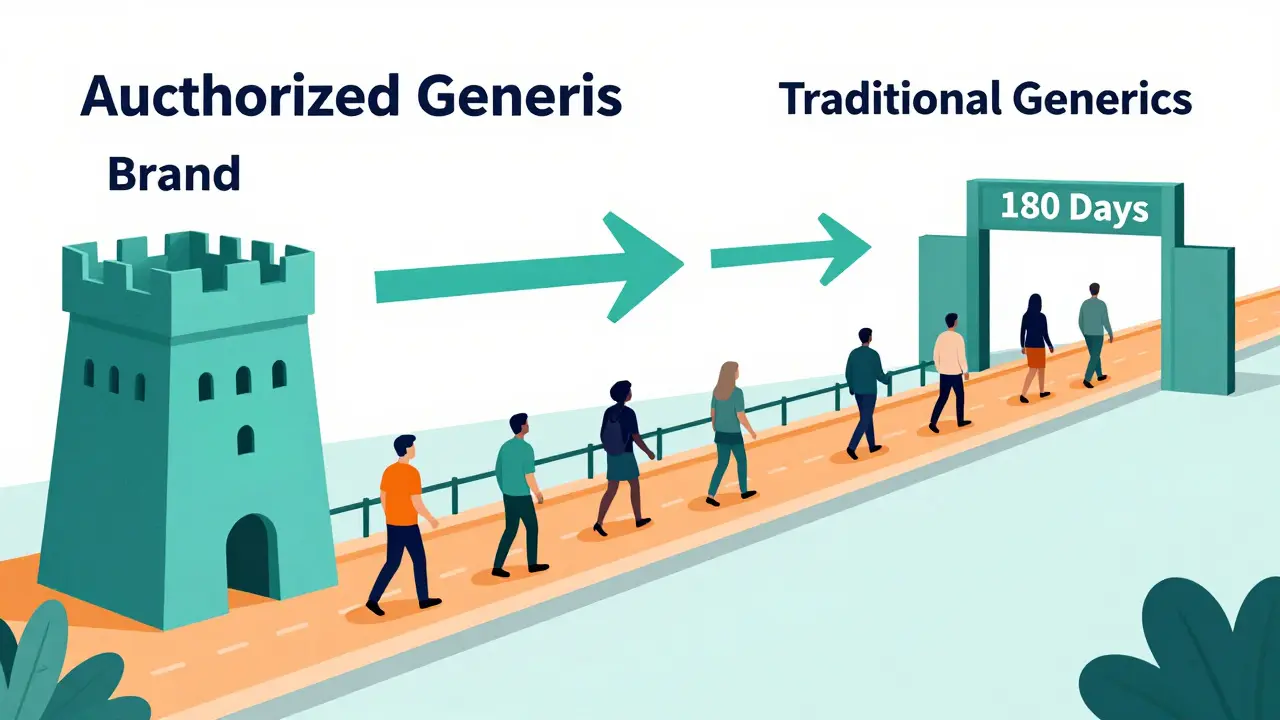
Price Sensitivity and the 180-Day Window
Money is always a factor in healthcare decisions. Authorized generics offer a moderate price reduction compared to brand-name drugs, typically lowering retail prices by 4-8% and wholesale prices by 7-14% during the critical first 180 days of generic competition. More recent data from 2022 shows that in some cases, such as with the drug entacapone, price reductions reached 13-18% in on-invoice costs.
However, here’s the catch: authorized generics are usually more expensive than traditional generics. Once the initial 180-day exclusivity period ends for the first generic competitor, traditional generics often drop to 30-50% lower prices than the brand. After this window, authorized generics typically remain 15-25% more expensive than traditional generics. So, while they offer immediate savings, they aren’t the cheapest option in the long run.
This creates a tiered preference system among patients. Those who prioritize cost above all else will eventually migrate to traditional generics once they become widely available. But patients who value formulation consistency and are less price-sensitive may stick with authorized generics, especially if their insurance covers them similarly.

The Role of Insurance and Pharmacy Benefit Managers
Here’s a reality check: most patients don’t actually get to choose between authorized generics and traditional generics. The choice is often made for them by their insurance companies and Pharmacy Benefit Managers (PBMs). According to a 2022 KFF analysis, 82% of commercial insurance plans automatically substitute generics for brand-name drugs when available. Only 12% allow patients or physicians to request the brand without prior authorization.
When a substitution occurs, the pharmacy dispensing software usually defaults to the lowest-cost alternative approved by the plan. If an authorized generic is listed as a preferred generic on the formulary, you’ll get that. If a traditional generic is preferred, you’ll get that. This means patient preference is often secondary to payer economics. However, pharmacists do have a role here. They can counsel patients on the differences, explaining that an authorized generic is made by the same company as the brand, which might alleviate concerns about efficacy or side effects.
Are Authorized Generics Anti-Competitive?
Not everyone views authorized generics positively. Critics argue that they undermine the intent of the Hatch-Waxman Act of 1984, which was designed to promote robust generic competition. By launching their own generics, brand companies can delay the entry of independent competitors. The FTC noted in 2011 that some brand companies use the threat of launching an authorized generic to induce generic firms to delay bringing their drugs to market. This practice, known as "pay-for-delay," can keep prices artificially high for longer.
Despite these concerns, authorized generics do provide immediate benefits to consumers. They introduce price competition earlier than traditional generics alone would. During the 180-day exclusivity period, authorized generics capture approximately 35-40% of the market share, forcing the brand name to lower its prices. Without them, the brand might maintain monopoly pricing for the entire exclusivity window. So, while they may hurt independent generic manufacturers, they often help patients save money in the short term.
How to Identify an Authorized Generic
If you’re curious whether your medication is an authorized generic, it can be tricky to tell. They look just like traditional generics on the shelf. The best way to identify them is through the FDA’s Orange Book. Authorized generics appear in the book as "products with no applicant" because they operate under the brand’s NDA rather than having their own ANDA approval number. Pharmacists and knowledgeable patients can check this list, updated monthly, to verify the source of their medication.
As of 2023, the FDA has drafted guidance to improve transparency around authorized generic identification, making it easier for consumers to know exactly what they are taking. Until then, asking your pharmacist is the most reliable method. They can tell you if the generic version you received was manufactured by the same company as the brand.
Future Trends in the Market
The landscape of authorized generics is evolving. Currently, they represent about $8-10 billion in annual U.S. pharmaceutical sales, growing at a rate of 5-7% per year. Major players like Pfizer, Merck, and AbbVie have launched authorized generics for various products, particularly in cardiovascular and central nervous system categories. Analysts predict that by 2028, authorized generics could account for 15-18% of the entire generic market.
However, regulatory scrutiny is increasing. The Congressional Budget Office warned in 2023 that if brand companies continue to use authorized generics to delay full generic competition, it could cost Medicare Part D beneficiaries $1.2 billion annually by 2027. This tension between immediate consumer savings and long-term market competition will likely shape future policies and patient access.
Is an authorized generic the same as a brand-name drug?
Yes, an authorized generic is chemically identical to the brand-name drug. It contains the same active and inactive ingredients and is manufactured by the same company in the same facilities. The only difference is the label; it does not carry the brand name.
Why are authorized generics more expensive than traditional generics?
Authorized generics are typically more expensive because they are introduced earlier in the market lifecycle, often during the 180-day exclusivity period for the first generic competitor. They also lack the intense price competition that arises when multiple third-party manufacturers enter the market later on.
Can my doctor prescribe an authorized generic specifically?
While doctors can write prescriptions for specific brands, most insurance plans automatically substitute generics. If you want an authorized generic, you may need to ask your pharmacist to check if it is available and covered by your plan. Some states allow "dispense as written" requests, but this is rare for generics.
Do authorized generics work differently than traditional generics?
No, they work the same way. Both authorized and traditional generics must meet FDA standards for safety and efficacy. However, because authorized generics use the exact same formulation as the brand, some patients report fewer issues with side effects or absorption compared to traditional generics, which may have different inactive ingredients.
How common are authorized generics in the U.S. market?
As of 2023, authorized generics account for approximately 12% of all generic drug units dispensed in the U.S. They are present in about 10-15% of drug categories experiencing generic competition, with growth expected to reach 15-18% of the generic market by 2028.